Research Laboratory Chemicals, New Processes, and Process Changes
Before the first time a Particularly Hazardous Substance (PHS) is used in your lab space, certain safety documentation and review requirements must be met. The two following lists sum up current PHSs at ASU:
EHS Particularly Hazardous Substances
FSE Particularly Hazardous Substances
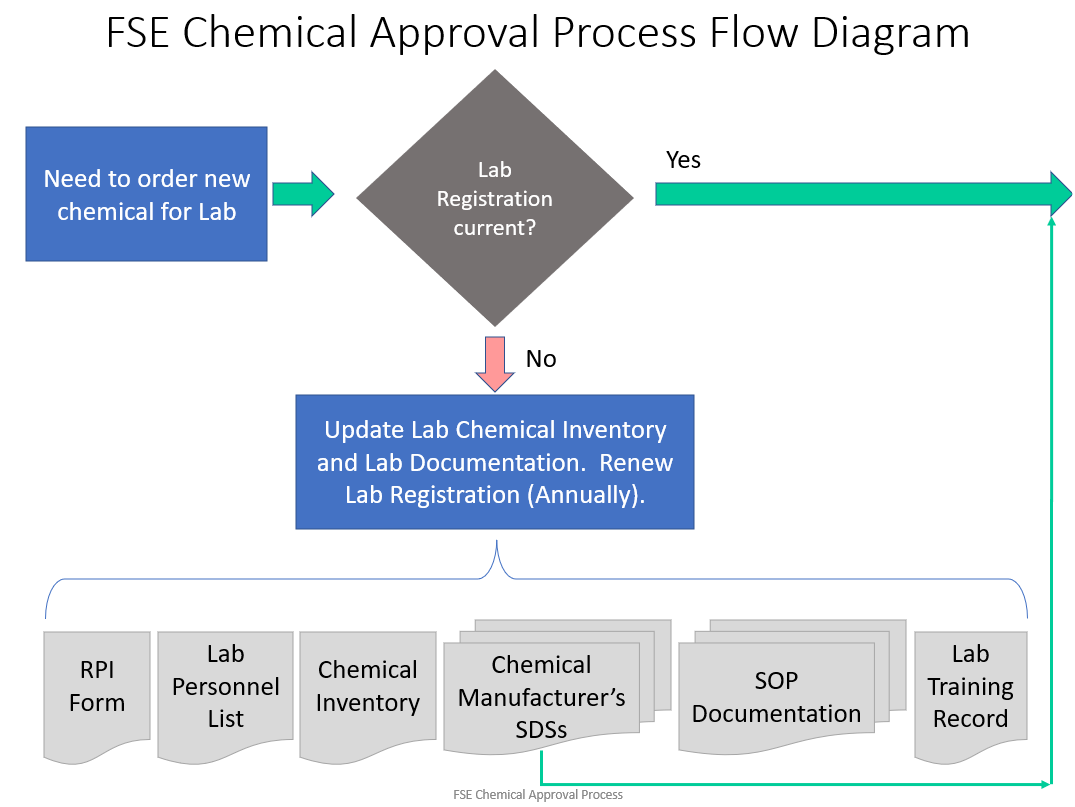
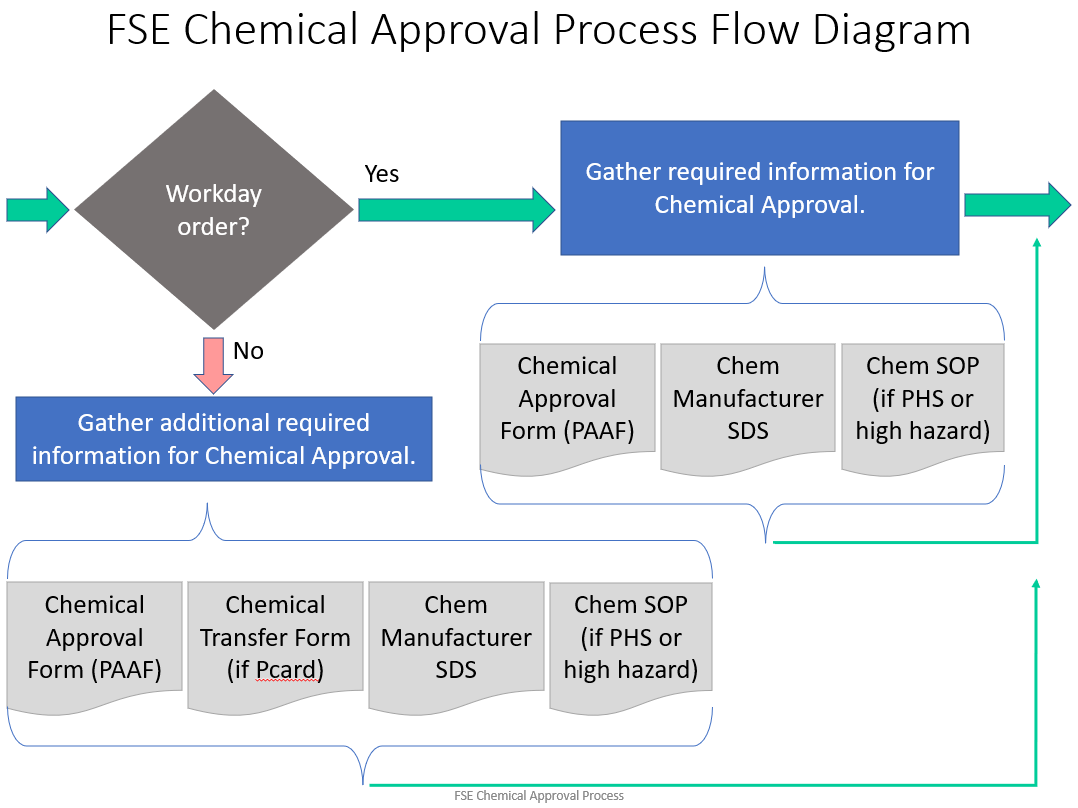
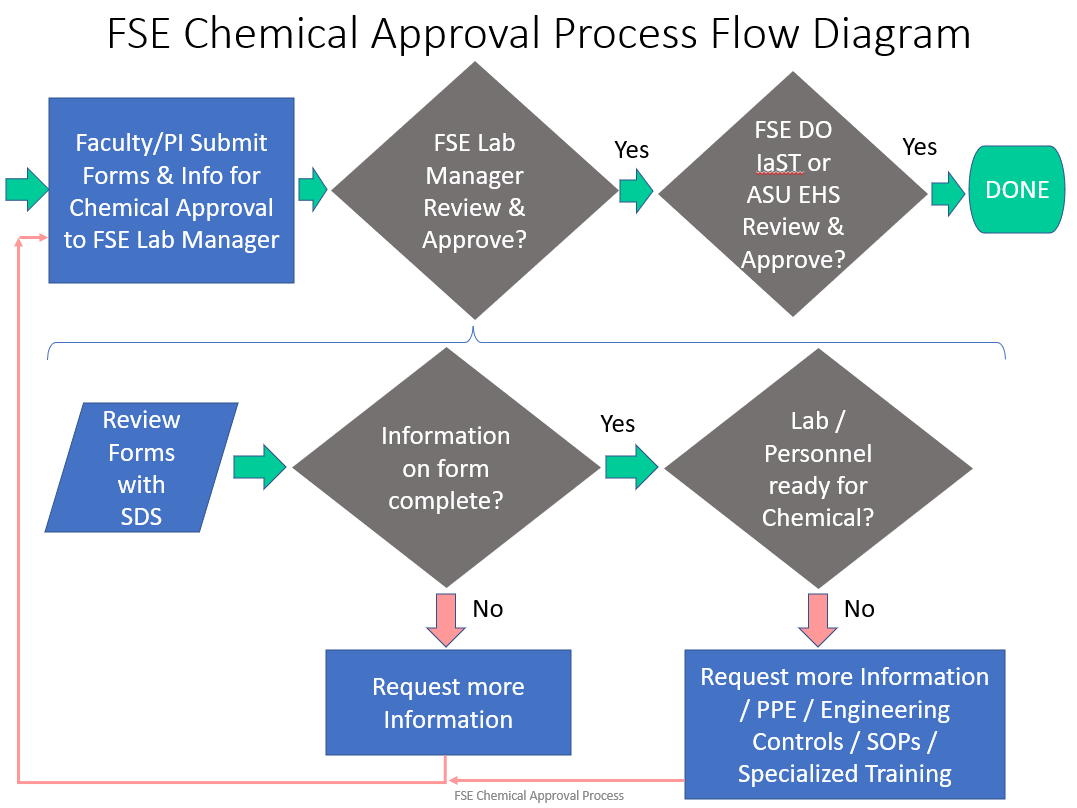
Any process that uses a PHS, and any process that uses a toxic or flammable gas, must follow the Chemical Approval Process before the process can be started and before equipment can be started up.
If a process has received approval before, and a change is made to the process, apparatus, or materials / concentration of materials, then the changes must be reviewed and approved before the process change can be implemented.
How to Receive Approval for a New Chemical, New Process, or for Changes to an Approved Process
Hazard Pages & Standard Operating Procedures (SOPs)
FSE’s existing resources for safely using a specific chemical or chemical hazard class, including SOP templates, are summed up on our Hazard Pages. Use the search bar below to search for a specific chemical, or use the slider to browse our database.
If a Hazard Page or chemical-specific SOP template has not yet been created for a chemical, use FSE’s general SOP templates to make something that fits your lab’s needs: